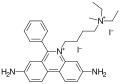
Propidium iodide
Propidium iodide (PI) is a fluorescent intercalating agent that can be used to stain cells. It is often used in Flow cytometry to evaluate cell viability or DNA content in cell cycle analysis.
Chemical Properties[edit]
Propidium iodide is a member of the Iodide family, with a molecular formula of C27H34I2N4. It has a molar mass of 668.4 g/mol. It is a red-orange compound that is soluble in water and has a melting point of 300 degrees Celsius.
Usage[edit]
Propidium iodide is used in Biotechnology and Molecular biology as a nucleic acid stain in a range of applications. It binds to DNA by intercalating between the bases with little or no sequence preference and with a stoichiometry of one dye per 4-5 base pairs of DNA. PI also binds to RNA, necessitating treatment of cells with RNase to ensure specific staining of DNA.
Flow Cytometry[edit]
In flow cytometry, propidium iodide is a popular DNA stain to assess cell cycle phase. It can be used in conjunction with other dyes that stain for different cellular components, such as Annexin V, which stains for apoptotic cells.
Safety[edit]
Propidium iodide is considered hazardous and should be handled with care. It is a potential mutagen and should be used with appropriate protective clothing and eyewear.
See Also[edit]
-
Propidium iodide
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian