Polyvinyl acetate
A synthetic polymer used in adhesives, paints, and coatings
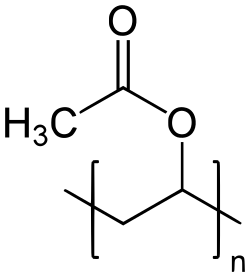
Polyvinyl acetate (PVA) is a synthetic polymer with the formula (C4H6O2)n. It is a type of vinyl polymer and is widely used in various industrial and consumer applications.
Properties[edit]
Polyvinyl acetate is a colorless, odorless, and tasteless solid. It is a thermoplastic polymer, meaning it becomes pliable or moldable above a specific temperature and solidifies upon cooling. PVA is known for its excellent adhesion properties, flexibility, and resistance to UV radiation.
Production[edit]
PVA is produced by the polymerization of vinyl acetate monomers. This process involves the use of free radical initiators to create long chains of polyvinyl acetate. The polymerization can be carried out in bulk, solution, or emulsion, with emulsion polymerization being the most common method.
Applications[edit]
Polyvinyl acetate is used in a variety of applications due to its adhesive properties and versatility.
Adhesives[edit]
PVA is a key ingredient in many types of adhesives, including white glue, wood glue, and school glue. Its ability to form strong bonds with porous materials makes it ideal for woodworking and paper products.
Paints and Coatings[edit]
In the paint industry, PVA is used as a binder in water-based paints. It helps improve the paint's adhesion to surfaces and enhances its durability. PVA is also used in coatings for textiles and paper.
Textiles[edit]
PVA is used in the textile industry as a sizing agent. It helps strengthen yarns and fabrics during weaving and reduces breakage.
Other Uses[edit]
PVA is also used in the production of polyvinyl alcohol and polyvinyl acetate emulsions, which have applications in the paper, textile, and packaging industries.
Environmental Impact[edit]
Polyvinyl acetate is considered to be a relatively environmentally friendly polymer. It is non-toxic and does not release harmful chemicals into the environment. However, like many synthetic polymers, it is not biodegradable and can contribute to plastic waste if not properly managed.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian