Phosphinate
Phosphinate[edit]
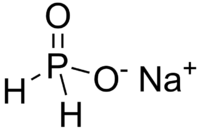
Phosphinates, also known as hypophosphites, are a class of chemical compounds containing the phosphinate ion, which is a derivative of phosphorous acid. These compounds are characterized by the presence of the P(=O)(OH)_ group, where phosphorus is bonded to two hydroxyl groups and one oxygen atom through a double bond.
Phosphinates are important in various industrial and chemical processes, including as reducing agents and in the synthesis of other phosphorus-containing compounds. They are also used in the formulation of flame retardants and as additives in electroless nickel plating.
Chemical Structure[edit]
The general structure of a phosphinate ion is represented as H_PO__. In this ion, the phosphorus atom is in the +1 oxidation state, and it is bonded to two hydroxyl groups and one oxygen atom. The presence of the P-H bond is a distinctive feature of phosphinates, differentiating them from phosphonates and phosphates.
Applications[edit]
Phosphinates are utilized in a variety of applications due to their unique chemical properties:
- Reducing Agents: Phosphinates are effective reducing agents in chemical reactions, particularly in the reduction of metal ions to their elemental state.
- Flame Retardants: Certain phosphinate compounds are used as flame retardants in polymers and textiles, providing fire resistance by promoting char formation and reducing flammability.
- Electroless Plating: Sodium hypophosphite is commonly used in electroless nickel plating, where it serves as a reducing agent to deposit nickel onto a substrate without the use of an external electrical current.
Synthesis[edit]
Phosphinates can be synthesized through several methods, including the reaction of phosphorus trichloride with water or alcohols, or by the hydrolysis of phosphonates. The choice of synthesis method depends on the desired phosphinate compound and its intended application.
Safety and Handling[edit]
Phosphinates, like many phosphorus-containing compounds, should be handled with care. They can be reactive and may release phosphine gas under certain conditions, which is toxic and flammable. Proper safety protocols, including the use of personal protective equipment and adequate ventilation, should be followed when working with phosphinates.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian