Phosphonate
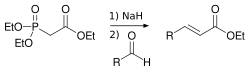
Phosphonates are a class of chemical compounds that contain the phosphonate (PO3−) functional group. They are used in many industrial applications, including water treatment, detergent manufacturing, and pharmaceutical production.
Chemical Structure[edit]
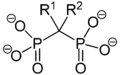
Phosphonates are characterized by a phosphorus atom bonded to three oxygen atoms and one carbon atom. This structure is similar to that of phosphates, but with a carbon atom replacing one of the oxygen atoms. This difference in structure gives phosphonates unique chemical properties, such as high stability and resistance to hydrolysis.
Uses[edit]
Phosphonates are used in a wide range of industrial applications. In water treatment, they are used to prevent the formation of scale and to control corrosion. In detergent manufacturing, they are used as builders to enhance the cleaning power of the detergent. In pharmaceutical production, they are used as intermediates in the synthesis of certain drugs.
Health and Environmental Effects[edit]
Phosphonates are generally considered to be safe for human exposure at low levels. However, they can be harmful if ingested or inhaled in large amounts. They can also cause skin and eye irritation. In the environment, phosphonates can contribute to the eutrophication of water bodies, leading to harmful algal blooms.
Gallery[edit]
-
Biphosphonate Structural Formulae
-
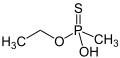
O-Ethyl methylphosphonothioic acid
-
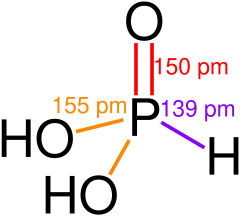
VX S-enantiomer
-
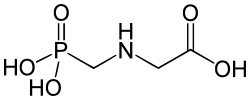
2-aminoethylphosphonate
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian