Phenyl group
Phenyl Group[edit]
The phenyl group is a common functional group in organic chemistry, consisting of a benzene ring with one hydrogen atom removed. It is represented by the symbol "Ph" or "C6H5" and is often used as a substituent in various organic compounds. The phenyl group plays a significant role in the structure and reactivity of many important organic molecules.
Structure[edit]
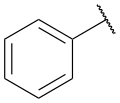
The phenyl group is composed of a six-membered carbon ring, with alternating single and double bonds. It is planar and exhibits aromaticity, which is a property of benzene and its derivatives. The delocalized π electrons in the benzene ring contribute to its stability and unique reactivity.
Properties[edit]
The presence of the phenyl group in a molecule can significantly influence its physical and chemical properties. Due to the aromatic nature of the benzene ring, compounds containing the phenyl group often exhibit enhanced stability and resistance to chemical reactions. Additionally, the phenyl group can affect the polarity and solubility of a molecule, depending on the substituents attached to it.
Applications[edit]
The phenyl group is widely used in various fields of chemistry and industry. It serves as a building block for the synthesis of numerous organic compounds, including pharmaceuticals, agrochemicals, and dyes. The presence of the phenyl group can impart specific properties to these compounds, such as increased lipophilicity or improved binding affinity to target receptors.
Reactions[edit]
The phenyl group participates in a variety of chemical reactions, allowing for the synthesis of diverse organic compounds. Some common reactions involving the phenyl group include electrophilic aromatic substitution, nucleophilic aromatic substitution, and oxidation reactions. These reactions can lead to the introduction of different functional groups onto the phenyl ring, expanding the range of possible derivatives.
Related Compounds[edit]
Several compounds are closely related to the phenyl group and share similar properties. These include the tolyl group (C6H4CH3), which is derived from the phenyl group by replacing one hydrogen atom with a methyl group, and the benzyl group (C6H5CH2), which contains an additional methylene group attached to the phenyl ring.
See Also[edit]
- Benzene - The parent compound of the phenyl group.
- Aromaticity - The property exhibited by the phenyl group due to the delocalized π electrons.
- Functional Group - A group of atoms responsible for the characteristic chemical reactions of a compound.
References[edit]
-
Phenyl group structure
-
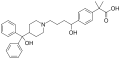
Atorvastatin molecule
-
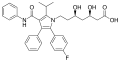
Fexofenadine structure
-
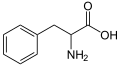
Phenylalanine structure
-
Bifenyl structure
-
Chlorobenzene structure
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian