Pertechnetate
Pertechnetate[edit]
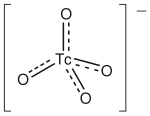
The pertechnetate ion is a chemical species with the formula TcO__. It is the most stable form of technetium in aqueous solution and is commonly encountered in the field of nuclear medicine. The pertechnetate ion is analogous to the permanganate ion, MnO__, and the perrhenate ion, ReO__, in terms of structure and charge.
Properties[edit]
Pertechnetate is a tetrahedral anion, meaning it has a central technetium atom surrounded by four oxygen atoms arranged in a tetrahedral geometry. This configuration is typical for high oxidation state metal oxoanions. The technetium in pertechnetate is in the +7 oxidation state, which is the highest oxidation state for technetium.
Pertechnetate is colorless and diamagnetic, and it is soluble in water. It is a weak oxidizing agent compared to permanganate, but it can still participate in redox reactions under certain conditions.
Applications[edit]
Pertechnetate is widely used in nuclear medicine as a radiopharmaceutical. The isotope technetium-99m (Tc-99m) is a metastable nuclear isomer used in various diagnostic imaging procedures. Tc-99m is obtained from a molybdenum-99 generator, where it is eluted as sodium pertechnetate (NaTcO_).
In medical imaging, Tc-99m pertechnetate is used for imaging the thyroid gland, salivary glands, and for brain imaging in certain cases. It is also used in renal imaging and to assess gastrointestinal bleeding.
Chemistry[edit]
In aqueous solution, pertechnetate is stable over a wide range of pH. It can be reduced to lower oxidation states of technetium, such as Tc(IV) and Tc(V), under reducing conditions. The reduction of pertechnetate is an important consideration in the management of radioactive waste, as technetium can be a long-lived fission product in nuclear reactors.
Safety[edit]
As a radioactive substance, pertechnetate must be handled with care to minimize exposure to radiation. In medical settings, the use of Tc-99m pertechnetate is carefully controlled to ensure patient and staff safety.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian