Technetium

Technetium is a chemical element with the symbol Tc and atomic number 43. It is the lightest element whose isotopes are all radioactive; none are stable. Nearly all technetium is produced synthetically, and it is primarily used in the field of nuclear medicine.
History[edit]
Technetium was the first element to be artificially produced. It was discovered in 1937 by Carlo Perrier and Emilio Segrè in a sample of molybdenum that had been bombarded with deuterons in a cyclotron. The element was named from the Greek word "technetos," meaning "artificial," reflecting its synthetic origin.
Properties[edit]
Technetium is a silvery-gray metal that tarnishes slowly in moist air. It is a member of the transition metals and is located in group 7 of the periodic table. Technetium is chemically similar to rhenium and manganese.
Isotopes[edit]

The most common isotope of technetium is technetium-99, which is a product of the fission of uranium-235 in nuclear reactors. Technetium-99m, a metastable nuclear isomer, is used in various diagnostic tests in nuclear medicine due to its short half-life of about 6 hours and its ability to emit gamma rays.
Applications[edit]
Technetium is primarily used in nuclear medicine for diagnostic imaging. The isotope technetium-99m is used in approximately 85% of all diagnostic imaging procedures in nuclear medicine. It is used in bone scans, myocardial perfusion imaging, and other diagnostic tests.
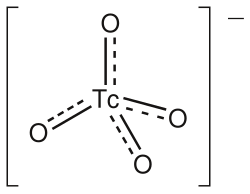
Chemical Compounds[edit]
Technetium forms a variety of chemical compounds, including oxides, halides, and complex ions. The pertechnetate ion (TcO₄⁻) is the most stable form of technetium in aqueous solutions and is used in many radiopharmaceuticals.

Production[edit]
Technetium is produced as a byproduct of the fission of uranium-235 in nuclear reactors. It can also be produced by bombarding molybdenum-98 with neutrons. The extraction of technetium from spent nuclear fuel involves complex chemical processes.
Safety[edit]
Technetium is radioactive, and its handling requires precautions to avoid exposure. The most common isotope, technetium-99, has a half-life of 211,000 years and decays by beta emission. Proper shielding and handling protocols are essential to ensure safety.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian