Paraformaldehyde
Paraformaldehyde[edit]
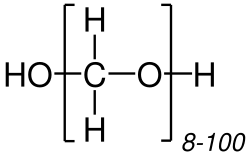
Paraformaldehyde is a polymer of formaldehyde with the chemical formula (CH_O)_, where n is typically 8-100. It is a white, crystalline solid that is used in various applications, particularly in biology and medicine as a fixative for tissue samples.
Chemical Properties[edit]
Paraformaldehyde is a polyacetal, which means it is a polymer formed from the repeated condensation of formaldehyde molecules. It is insoluble in water and has a characteristic formaldehyde odor. Upon heating, paraformaldehyde depolymerizes to release formaldehyde gas, which is the basis for its use as a fumigant and disinfectant.
Uses[edit]
Paraformaldehyde is widely used in histology and pathology for the fixation of biological tissues. It is preferred over formaldehyde solutions because it can be stored as a solid and dissolved in water to produce formaldehyde solutions of desired concentrations when needed.
Fixation in Histology[edit]
In histological applications, paraformaldehyde is used to preserve tissue samples by cross-linking proteins, which stabilizes the tissue structure and prevents degradation. This process is crucial for preparing samples for microscopy and other analytical techniques.
Disinfectant and Fumigant[edit]
Paraformaldehyde is also used as a disinfectant and fumigant due to its ability to release formaldehyde gas upon heating. This property makes it useful for sterilizing equipment and surfaces in laboratories and medical facilities.
Safety and Handling[edit]
Paraformaldehyde is a hazardous material and should be handled with care. It can release formaldehyde gas, which is a known carcinogen and can cause irritation to the respiratory system, skin, and eyes. Proper personal protective equipment (PPE) such as gloves, goggles, and masks should be used when handling paraformaldehyde.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian