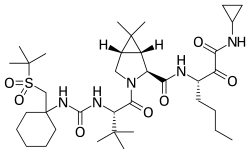
Narlaprevir
Overview[edit]
Narlaprevir is an antiviral drug used in the treatment of hepatitis C. It is a protease inhibitor that targets the NS3/4A serine protease of the hepatitis C virus (HCV), thereby inhibiting viral replication. Narlaprevir is primarily used in combination with other antiviral agents to enhance its efficacy.
Mechanism of Action[edit]
Narlaprevir functions by binding to the active site of the NS3/4A serine protease, an enzyme critical for the viral replication process of HCV. By inhibiting this enzyme, narlaprevir prevents the cleavage of the HCV polyprotein into functional viral proteins, thereby halting the replication of the virus within the host cells.
Pharmacokinetics[edit]
Narlaprevir is administered orally and undergoes extensive hepatic metabolism. It is primarily metabolized by the cytochrome P450 enzyme system, particularly CYP3A4. The drug and its metabolites are excreted mainly via the biliary system and to a lesser extent through the renal system.
Clinical Use[edit]
Narlaprevir is used in the treatment of chronic hepatitis C infection, particularly in patients infected with HCV genotype 1. It is often used in combination with other antiviral agents such as ribavirin and pegylated interferon to improve treatment outcomes. The combination therapy aims to achieve a sustained virologic response (SVR), which is indicative of the virus being undetectable in the blood 12 weeks after completing treatment.
Side Effects[edit]
Common side effects of narlaprevir include fatigue, nausea, headache, and anemia. More serious adverse effects can occur, such as liver enzyme elevation and rash. Patients receiving narlaprevir should be monitored for these potential side effects, and dose adjustments may be necessary.
Drug Interactions[edit]
Narlaprevir is known to interact with other drugs metabolized by the CYP3A4 enzyme. Caution should be exercised when co-administering narlaprevir with other medications that are strong inducers or inhibitors of CYP3A4, as these can alter the plasma concentration of narlaprevir, potentially affecting its efficacy and safety.
Development and Approval[edit]
Narlaprevir was developed as part of the effort to improve treatment options for hepatitis C, particularly for patients who do not respond well to traditional therapies. It has been approved for use in several countries and continues to be a part of ongoing research to optimize hepatitis C treatment regimens.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian