N-Methylethanolamine
N-Methylethanolamine (NMEA) is an organic compound with the formula CH3NHCH2CH2OH. It is a colorless liquid with an ammonia-like odor and is a member of the ethanolamines, compounds that contain both an amine and an alcohol functional group in the same molecule. N-Methylethanolamine is used in a variety of chemical synthesis processes and has applications in different industries, including pharmaceuticals, agriculture, and chemical manufacturing.
Properties[edit]
N-Methylethanolamine has a molecular weight of 75.11 g/mol and a boiling point of approximately 160°C. It is soluble in water, which makes it useful in aqueous solutions for various chemical reactions and applications. Its dual functionality as both an amine and an alcohol allows it to participate in a wide range of chemical reactions, making it a versatile intermediate in organic synthesis.
Synthesis[edit]
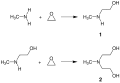
N-Methylethanolamine is typically synthesized through the reaction of ethylene oxide with methylamine. This process involves the nucleophilic attack of the amine on the ethylene oxide, resulting in the opening of the epoxide ring and formation of the ethanolamine derivative. The reaction conditions, such as temperature, pressure, and the ratio of reactants, can be adjusted to optimize the yield and purity of N-Methylethanolamine.
Applications[edit]
Pharmaceuticals[edit]
In the pharmaceutical industry, N-Methylethanolamine is used as a building block for the synthesis of active pharmaceutical ingredients (APIs). Its ability to act as a chelating agent, due to the presence of both amine and alcohol functional groups, makes it useful in the development of certain drugs.
Agriculture[edit]
N-Methylethanolamine serves as a precursor in the synthesis of plant growth regulators and pesticides. Its effectiveness in these applications is attributed to its chemical properties, which allow it to interact with biological molecules in plants.
Chemical Manufacturing[edit]
In chemical manufacturing, N-Methylethanolamine is used in the production of surfactants, emulsifiers, and corrosion inhibitors. Its amphiphilic nature, owing to the presence of both hydrophilic and hydrophobic functional groups, makes it suitable for applications requiring the stabilization of mixtures of oil and water.
Safety and Handling[edit]
N-Methylethanolamine should be handled with care, as it can cause irritation to the skin, eyes, and respiratory system. Proper personal protective equipment (PPE), such as gloves and goggles, should be worn when handling this chemical. Additionally, it should be stored in a cool, well-ventilated area away from sources of ignition.
Environmental Impact[edit]
The environmental impact of N-Methylethanolamine depends on its concentration and the context of its release into the environment. As with many chemicals, it is important to follow proper disposal procedures and regulatory guidelines to minimize any potential harm to the environment.
-
Synthesis of N-Methylethanolamine and MDEA
-
Synthesis of Aziridin
-
DNA Repair Mechanism
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian