Methyl isobutyrate
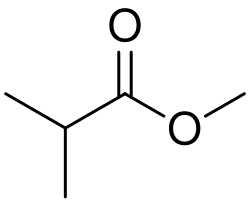
Methyl isobutyrate, also known as methyl 2-methylpropanoate, is an organic compound with the molecular formula C_5H_10O_2. It is a colorless liquid that is used in the flavor and fragrance industry due to its fruity odor, reminiscent of apples or pineapples. This compound is found in small amounts in various fruits and is used to impart or enhance fruity flavors in food products, beverages, and cosmetics.
Properties[edit]
Methyl isobutyrate has a boiling point of 102 °C and a density of 0.897 g/cm^3 at 20 °C. It is slightly soluble in water, but it is well soluble in most organic solvents such as ethanol, diethyl ether, and chloroform. The compound's fruity odor and flavor make it a valuable additive in the production of food flavorings and fragrances.
Synthesis[edit]
Methyl isobutyrate can be synthesized through the esterification of isobutyric acid (2-methylpropanoic acid) with methanol. This reaction typically involves the use of an acid catalyst, such as sulfuric acid, to facilitate the formation of the ester bond.
Applications[edit]
The primary use of methyl isobutyrate is in the flavor and fragrance industry. It is added to a variety of food products, including candies, baked goods, and beverages, to impart a fruity flavor. In the fragrance industry, it is used in the formulation of perfumes and scents to provide a fresh, fruity aroma.
Safety[edit]
Methyl isobutyrate is generally considered safe for use in food and cosmetic products. However, like many organic solvents, it should be handled with care as it can cause irritation to the skin and eyes upon contact. Adequate ventilation should be ensured when working with this compound to avoid inhalation of vapors.
Environmental Impact[edit]
While methyl isobutyrate is not known to be a major environmental pollutant, its production and use should be managed to minimize release into the environment. Proper disposal and recycling procedures should be followed to prevent contamination of water and soil.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
