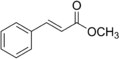
Methyl cinnamate
Methyl cinnamate is an organic compound that is commonly used in the flavor and fragrance industry due to its pleasant aroma, which is similar to that of strawberry. It is a member of the ester family, which are compounds produced by the reaction between acids and alcohols. Methyl cinnamate is derived from cinnamic acid, and its chemical formula is C10H10O2.
Chemical Structure[edit]
Methyl cinnamate consists of a benzene ring attached to a carboxyl group (COOCH3). The benzene ring contributes to the compound's aromaticity, while the carboxyl group is responsible for its ester properties. The presence of the methyl group (CH3) in the carboxyl group gives the compound its name.
Production[edit]
Methyl cinnamate is typically produced through the esterification of cinnamic acid and methanol, in the presence of a strong acid such as sulfuric acid. This reaction results in the formation of methyl cinnamate and water.
Uses[edit]
Due to its sweet, fruity aroma, methyl cinnamate is widely used in the flavor and fragrance industry. It is often used in the formulation of perfumes and cosmetics, and as a flavoring agent in food and beverages. In addition, it has been found to exhibit antimicrobial properties, making it useful in the preservation of certain food products.
Safety[edit]
While generally considered safe for use in food and cosmetics, methyl cinnamate can cause allergic reactions in some individuals. It is therefore important to use this compound with caution, particularly in products that are intended for topical application.
See Also[edit]
-
Methyl cinnamate
-
Methyl cinnamate 3D spacefill
-
Methyl cinnamate
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian