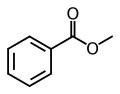
Methyl benzoate
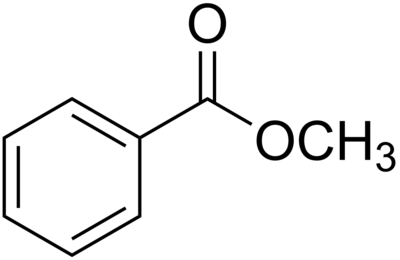
Methyl benzoate is an organic compound with the chemical formula C7H6O2. It is a methyl ester derived from benzoic acid. This colorless liquid is sparingly soluble in water, but miscible with organic solvents. It has a pleasant odor, reminiscent of some fruits and flowers, and is used in perfumery and as a flavoring agent. Methyl benzoate is also employed as a solvent and an intermediate in the synthesis of other chemical compounds.
Properties[edit]
Methyl benzoate has a melting point of -12.5 °C and a boiling point of 199.6 °C. Its density is 1.094 g/cm3. The compound is less dense than water, which allows it to float on water's surface. It has a sweet, floral scent, which has been described as similar to that of ylang-ylang or jasmine.
Synthesis[edit]
Methyl benzoate can be synthesized through the esterification reaction of benzoic acid with methanol, in the presence of a strong acid catalyst such as sulfuric acid or hydrochloric acid. This process is known as Fischer esterification. The reaction is reversible, and the yield can be improved by removing the water formed during the reaction.
Uses[edit]
Methyl benzoate is widely used in the flavor and fragrance industry due to its pleasant aroma. It is a component of various flower and fruit flavors and fragrances, used in perfumes, soaps, and cosmetics. In addition to its use in the consumer goods industry, methyl benzoate serves as a solvent for the synthesis of other organic compounds. It is also used in the preparation of certain dyes and pharmaceuticals.
Safety[edit]
Methyl benzoate is considered to be moderately toxic. It can cause irritation to the skin, eyes, and respiratory system upon exposure. Proper handling and protective equipment are recommended when working with this chemical. It is also important to work in a well-ventilated area to avoid inhalation of vapors.
Environmental Impact[edit]
While methyl benzoate is not considered a major environmental pollutant, it can contribute to the organic content of wastewater and should be disposed of properly. Biodegradation in soil and water is possible, leading to its breakdown into less harmful substances.
-
Methyl benzoate
-
Methyl benzoate 3D computer model
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian