Lithium fluoride
Lithium fluoride (LiF) is an inorganic compound with the chemical formula LiF. It is a white, crystalline solid that is highly stable and has a high melting point. Lithium fluoride is notable for its use in specialized optics and as a component in molten salt reactors.
Properties[edit]
Lithium fluoride is a crystalline solid with a high melting point of 845 °C. It has a density of 2.635 g/cm³ and is highly stable, making it resistant to decomposition. The compound is soluble in water, with a solubility of 0.27 g/100 mL at 18 °C. It is also slightly soluble in ethanol.
Structure[edit]
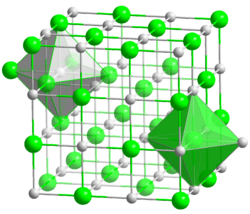
Lithium fluoride adopts the rock salt structure, which is a common structure for many ionic compounds. In this structure, each lithium ion (Li⁺) is surrounded by six fluoride ions (F⁻), and each fluoride ion is surrounded by six lithium ions. This arrangement results in a highly stable lattice.
Production[edit]
Lithium fluoride can be produced by the reaction of lithium hydroxide (LiOH) or lithium carbonate (Li₂CO₃) with hydrofluoric acid (HF): \[ \text{LiOH} + \text{HF} \rightarrow \text{LiF} + \text{H}_2\text{O} \] \[ \text{Li}_2\text{CO}_3 + 2\text{HF} \rightarrow 2\text{LiF} + \text{CO}_2 + \text{H}_2\text{O} \]
Applications[edit]
Optics[edit]
Lithium fluoride is used in specialized optics due to its transparency to ultraviolet (UV) light. It is used in UV and vacuum ultraviolet (VUV) optics, including lenses and windows. Its low refractive index and high transmission in the UV range make it valuable in these applications.
Nuclear Industry[edit]
In the nuclear industry, lithium fluoride is a component of molten salt reactors (MSRs). It is used in the form of LiF-BeF₂ (FLiBe) mixtures, which serve as a coolant and a solvent for nuclear fuel. The high thermal stability and low neutron absorption cross-section of lithium fluoride make it suitable for this purpose.
Other Uses[edit]
Lithium fluoride is also used in the production of ceramics, glass, and aluminum. It acts as a flux in the smelting of aluminum and in the production of certain types of glass and ceramics.
Safety[edit]
Lithium fluoride is considered to be of low toxicity, but it should be handled with care. Inhalation or ingestion of lithium fluoride can cause irritation to the respiratory and gastrointestinal tracts. Proper safety measures, including the use of personal protective equipment, should be followed when handling this compound.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
