Isomaltol
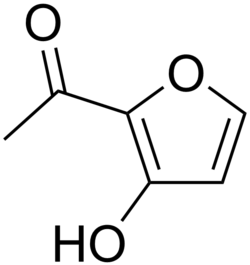
Isomaltol is a naturally occurring organic compound that belongs to the class of furan derivatives. It is a white crystalline solid with a sweet odor and is known for its flavor-enhancing properties. Isomaltol is commonly found in caramel and coffee, contributing to their characteristic flavors.
Chemical Structure and Properties[edit]
Isomaltol has the chemical formula C6H6O3 and a molecular weight of 126.11 g/mol. Its structure consists of a furan ring with a hydroxyl group and a methyl group attached to it. The IUPAC name for isomaltol is 3-hydroxy-2-methyl-4H-pyran-4-one.
Synthesis[edit]
Isomaltol can be synthesized through the Maillard reaction, which involves the reaction between reducing sugars and amino acids. This reaction is responsible for the browning and flavor development in various cooked foods. Additionally, isomaltol can be produced by the thermal degradation of starch and cellulose.
Applications[edit]
Isomaltol is widely used in the food industry as a flavor enhancer. It is commonly added to baked goods, confectionery, and beverages to impart a sweet, caramel-like flavor. In addition to its use in food, isomaltol has potential applications in the pharmaceutical industry due to its antioxidant properties.
Health and Safety[edit]
Isomaltol is generally recognized as safe (GRAS) by the Food and Drug Administration (FDA) when used in accordance with good manufacturing practices. However, excessive consumption of isomaltol-containing products may lead to adverse health effects.
Related Compounds[edit]
Isomaltol is structurally related to other furan derivatives such as maltol and ethyl maltol. These compounds also possess flavor-enhancing properties and are used in various food and beverage applications.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian