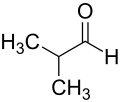
Isobutyraldehyde
Isobutyraldehyde is an organic compound with the formula (CH3)2CHCHO. It is a colorless liquid that is used in the manufacture of a variety of other chemicals.
Chemical Structure[edit]
Isobutyraldehyde is a four-carbon compound that is classified as an aldehyde. It has two methyl groups attached to a carbon atom, which is then attached to a carbon atom with a double bond to an oxygen atom. This structure is what gives isobutyraldehyde its characteristic properties.
Properties[edit]
Isobutyraldehyde is a colorless liquid with a strong, pungent smell. It is less dense than water and its vapors are heavier than air. It is highly flammable and can react with oxidizing agents.
Uses[edit]
Isobutyraldehyde is used as a precursor to other chemicals. It is used in the manufacture of isobutylamine, which is used in the production of isobutyl acetate. Isobutyraldehyde is also used in the synthesis of neopentyl glycol, a key ingredient in certain types of polyester resins and in synthetic lubricants. In the pharmaceutical industry, it is used as a building block in the synthesis of a variety of drugs.
Health Effects[edit]
Exposure to isobutyraldehyde can cause irritation to the eyes, skin, and respiratory tract. It can also cause nausea, headache, and dizziness. Long-term exposure can lead to more serious health effects, including damage to the liver and kidneys.
Safety[edit]
Due to its flammability and potential health effects, isobutyraldehyde should be handled with care. It should be stored in a cool, well-ventilated area away from sources of ignition. Protective clothing and eye protection should be worn when handling this chemical.
See Also[edit]
Isobutyraldehyde[edit]
-
Isobutyraldehyde
-
Pantothenic acid synthesis
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian