Iron(II) sulfate
Chemical compound
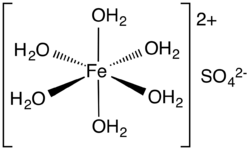
Iron(II) sulfate, also known as ferrous sulfate, is a chemical compound with the formula FeSO₄. It is commonly encountered as the heptahydrate, FeSO₄·7H₂O, which is a blue-green crystalline solid. This compound is used in various applications, including as a precursor to other iron compounds, in water treatment, and as a nutritional supplement.
Chemical Properties[edit]
Iron(II) sulfate is a sulfate salt of iron in the +2 oxidation state. The anhydrous form is a white crystalline solid, while the heptahydrate form is blue-green. The compound is soluble in water, and its solutions are acidic due to hydrolysis.

Hydration States[edit]
Iron(II) sulfate exists in several hydrated forms, the most common being the heptahydrate. Other hydrates include the monohydrate and tetrahydrate. The heptahydrate loses water upon heating, eventually forming the anhydrous form.
Production[edit]
Iron(II) sulfate is produced by treating iron with sulfuric acid, often as a byproduct of the pickling of steel:
- Fe + H₂SO₄ → FeSO₄ + H₂
It can also be obtained by the oxidation of pyrite (iron sulfide) or by the reaction of iron with copper(II) sulfate.
Applications[edit]
Industrial Uses[edit]
Iron(II) sulfate is used in the manufacture of iron(III) oxide pigments, as a reducing agent in the treatment of chromate-containing effluents, and in the production of iron(III) sulfate.

Agriculture[edit]
In agriculture, iron(II) sulfate is used as a soil amendment for lowering the pH of a high alkaline soil so that plants can access the soil's nutrients. It is also used to treat iron chlorosis, a condition in which leaves produce insufficient chlorophyll.
Medical Uses[edit]
Iron(II) sulfate is used as a dietary supplement to treat iron-deficiency anemia. It is available in various forms, including tablets and liquid solutions.
Safety[edit]
Iron(II) sulfate is generally considered safe when used as directed. However, it can be toxic in large amounts, particularly to children. It should be stored out of reach of children and handled with care to avoid ingestion.

Related Compounds[edit]
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian