Imidafenacin
Overview of the antimuscarinic drug Imidafenacin
| Imidafenacin | |
|---|---|
|
| |
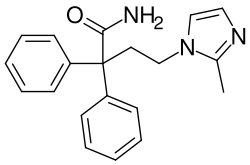
| IUPAC name | 4-(2-methyl-1H-imidazol-1-yl)-2,2-diphenylbutanamide
|
Imidafenacin is a medication used primarily in the treatment of overactive bladder (OAB). It is classified as an antimuscarinic agent, which works by inhibiting the action of acetylcholine on muscarinic receptors in the bladder, thereby reducing urinary urgency, frequency, and incontinence.
Pharmacology[edit]
Imidafenacin is a selective antagonist for the M3 and M1 subtypes of muscarinic receptors. These receptors are predominantly found in the bladder and salivary glands. By blocking these receptors, imidafenacin reduces involuntary bladder contractions and increases bladder capacity.
Mechanism of Action[edit]
The mechanism of action of imidafenacin involves competitive inhibition of muscarinic receptors. This inhibition decreases the detrusor muscle contractions of the bladder, which are responsible for the symptoms of overactive bladder. The selectivity for M3 receptors helps in minimizing side effects such as dry mouth and constipation, which are common with non-selective antimuscarinics.
Clinical Use[edit]
Imidafenacin is indicated for the management of overactive bladder with symptoms of urgency, frequency, and urge incontinence. It is administered orally and is available in tablet form. The typical dosage is 0.1 mg twice daily, but the dosage may be adjusted based on patient response and tolerability.
Side Effects[edit]
Common side effects of imidafenacin include dry mouth, constipation, and blurred vision. These effects are due to its anticholinergic properties. Less common side effects may include urinary retention, dizziness, and headache.
Contraindications[edit]
Imidafenacin is contraindicated in patients with urinary retention, gastric retention, uncontrolled narrow-angle glaucoma, and in those who have demonstrated hypersensitivity to the drug or its components.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian