Idose
Idose[edit]
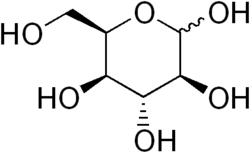
Idose is a monosaccharide and an aldohexose sugar. It is one of the eight aldohexose stereoisomers and is classified as a rare sugar. Idose is not commonly found in nature but is an important component in the structure of certain glycosaminoglycans, such as dermatan sulfate.
Structure[edit]
Idose has the chemical formula C_H__O_. It contains six carbon atoms, twelve hydrogen atoms, and six oxygen atoms. The structure of idose is characterized by the presence of an aldehyde group at the first carbon (C1) and hydroxyl groups attached to the other carbon atoms. The specific configuration of the hydroxyl groups distinguishes idose from other aldohexoses.
Stereochemistry[edit]
Idose has four chiral centers, which means it can exist in multiple stereoisomeric forms. The D- and L- forms of idose are enantiomers, meaning they are mirror images of each other. The D-form is the one most commonly referenced in biochemical contexts.
Biological Role[edit]
Idose is not typically found in free form in nature. However, it is a component of certain glycosaminoglycans, which are long unbranched polysaccharides consisting of repeating disaccharide units. In particular, idose is found in dermatan sulfate, where it alternates with N-acetylgalactosamine in the polysaccharide chain.
Synthesis[edit]
Idose can be synthesized in the laboratory through the epimerization of D-glucose or D-galactose. This process involves the alteration of the configuration of the hydroxyl group at the C5 position to produce idose.
Applications[edit]
While idose itself is not widely used in industrial applications, its derivatives and related compounds are of interest in the field of biochemistry and pharmacology. The study of idose and its derivatives can provide insights into the structure and function of glycosaminoglycans and their role in biological processes.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian