Hydroxybutyric acid

Chemical compound
| Hydroxybutyric acid | |
|---|---|
| Hydroxybutyric acid.svg | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
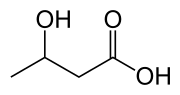
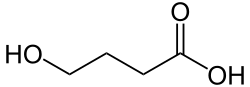
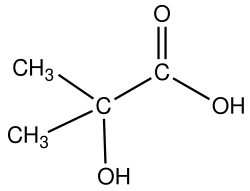
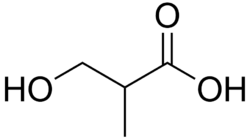
Hydroxybutyric acid (HBA) is a chemical compound with the molecular formula C4H8O3. It is a hydroxy acid and is classified as a carboxylic acid. The compound exists in several isomeric forms, including gamma-hydroxybutyric acid (GHB), which is a naturally occurring substance found in the human central nervous system, as well as in wine, beef, small citrus fruits, and almost all animals in small amounts.
Isomers[edit]
Hydroxybutyric acid has several isomers, including:
- gamma-Hydroxybutyric acid (GHB)
- beta-Hydroxybutyric acid (BHB)
- alpha-Hydroxybutyric acid (AHB)
Biological Role[edit]
Gamma-hydroxybutyric acid (GHB) is a neurotransmitter and a psychoactive drug. It is used medically as a general anesthetic and to treat conditions such as narcolepsy and alcoholism. GHB is also known for its use as a recreational drug and has been associated with cases of drug-facilitated sexual assault.
Synthesis[edit]
Hydroxybutyric acid can be synthesized through various chemical processes. One common method involves the reduction of gamma-butyrolactone (GBL) with a reducing agent such as sodium borohydride.
Metabolism[edit]
In the human body, hydroxybutyric acid is metabolized into carbon dioxide and water through the citric acid cycle. It is also a precursor to the synthesis of gamma-aminobutyric acid (GABA), a major inhibitory neurotransmitter in the central nervous system.
Applications[edit]
Hydroxybutyric acid and its derivatives have several applications:
- GHB is used in medicine for anesthesia and the treatment of narcolepsy.
- BHB is used as a marker for ketosis in metabolic studies.
- AHB is used in biochemical research.
Safety and Legal Status[edit]
The legal status of hydroxybutyric acid varies by country. In many places, GHB is classified as a controlled substance due to its potential for abuse and its association with drug-facilitated crimes.
See also[edit]
- gamma-Hydroxybutyric acid
- beta-Hydroxybutyric acid
- alpha-Hydroxybutyric acid
- Neurotransmitter
- Carboxylic acid
- Psychoactive drug
- General anesthetic
- Narcolepsy
- Alcoholism
- Drug-facilitated sexual assault
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian