Hexaethyl tetraphosphate
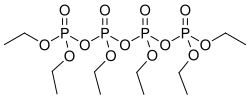
0Hexaethyl tetraphosphate is a chemical compound with the chemical formula C12H30O7P2. It is a type of organophosphate, a class of compounds that contain phosphoric acid esters. Hexaethyl tetraphosphate is primarily used as a flame retardant and plasticizer in the production of polyvinyl chloride (PVC) and other polymers.
Chemical Structure[edit]
Hexaethyl tetraphosphate consists of two phosphate groups bonded to four ethyl groups. The phosphate groups are connected by an oxygen atom, forming a P-O-P bond. This structure is characteristic of organophosphates, which are known for their high reactivity and potential for chemical interactions with other substances.
Properties[edit]
Hexaethyl tetraphosphate is a colorless, oily liquid at room temperature. It has a high boiling point and is relatively stable under normal conditions. However, it can decompose under high heat or in the presence of strong acids or bases. As an organophosphate, it has the potential to inhibit the activity of certain enzymes, although its toxicity to humans is relatively low.
Uses[edit]
The primary use of hexaethyl tetraphosphate is as a flame retardant and plasticizer in the production of polymers. It can be added to PVC and other materials to increase their flexibility and resistance to fire. It is also used in the production of certain types of resins and coatings.
Safety[edit]
Like other organophosphates, hexaethyl tetraphosphate can be harmful if ingested or inhaled. It can cause irritation to the skin and eyes, and prolonged exposure can lead to more serious health effects. Proper safety measures should be taken when handling this substance.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
