Hexachlorocyclohexane

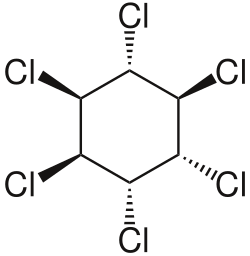
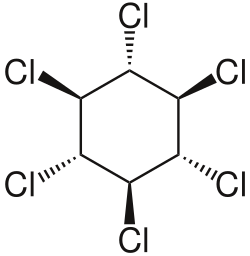
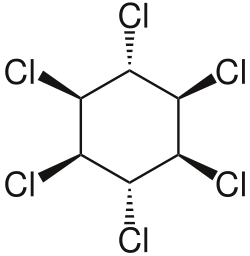
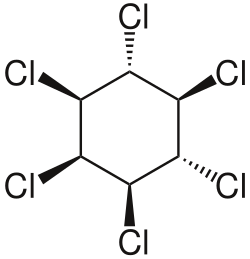
Hexachlorocyclohexane (HCH) is an organic compound that belongs to the family of chlorinated hydrocarbons. It is best known for its use as an insecticide. The chemical formula for hexachlorocyclohexane is C6H6Cl6. This compound exists in eight stereoisomers, among which the gamma-isomer, known as lindane, is the most active as an insecticide.
History[edit]
Hexachlorocyclohexane was first synthesized in the early 19th century, but its insecticidal properties were not discovered until the 1940s. Following this discovery, it was widely used to control agricultural pests and mosquitoes for malaria control. However, due to concerns over its environmental persistence, bioaccumulation, and potential to cause adverse health effects, its use has been significantly restricted and banned in many countries since the late 20th century.
Chemical Properties[edit]
Hexachlorocyclohexane is a colorless solid under normal conditions. It is poorly soluble in water but highly soluble in many organic solvents. The gamma-isomer, lindane, is the most well-known and widely studied due to its insecticidal properties. The other isomers, while similar in chemical structure, are less effective as pesticides and have different physical and chemical properties.
Uses[edit]
Historically, hexachlorocyclohexane was used in agriculture to control a wide range of pests. Lindane, the gamma-isomer, was also used in pharmaceutical products for treating lice and scabies. However, its use in agriculture and pharmaceuticals has been phased out or banned in many jurisdictions due to health and environmental concerns.
Health and Environmental Impact[edit]
Hexachlorocyclohexane is considered to be a persistent organic pollutant (POP). It can persist in the environment for long periods, bioaccumulate in the food chain, and pose risks to human health and the environment. Exposure to high levels of HCH can lead to neurological effects, liver damage, and possible links to cancer. Its persistence in the environment can also harm wildlife, particularly species at the top of the food chain.
Regulation[edit]
Due to its adverse effects, the production and use of hexachlorocyclohexane, particularly lindane, have been restricted under various international agreements, including the Stockholm Convention on Persistent Organic Pollutants. Many countries have implemented regulations to limit the use and disposal of HCH and its isomers.
Conclusion[edit]
While hexachlorocyclohexane played a significant role in pest control during the 20th century, its environmental and health impacts have led to a reevaluation of its use. Today, the focus has shifted towards finding safer and more sustainable alternatives for pest management and disease control.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
