Haloxon
| Haloxon | |
|---|---|
|
| |
| IUPAC name | IUPAC name of Haloxon
|
| CAS Number | 321-55-1 |
| PubChem | 16683
|
| ChemSpider | 15819
|
Haloxon is an organophosphate compound that has been used as an anthelmintic agent, particularly in veterinary medicine. It is primarily used to treat parasitic infections in animals, such as helminthiasis in livestock.
Chemical Structure and Properties[edit]
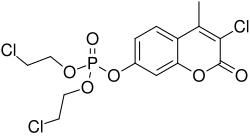
Haloxon is characterized by its organophosphate structure, which includes a phosphorus atom bonded to an oxygen atom and a chlorine atom. The chemical formula for Haloxon is C₉H₁₄ClO₄P, and it has a molecular weight of approximately 252.63 g/mol.
Mechanism of Action[edit]
Haloxon functions by inhibiting the enzyme acetylcholinesterase, which is crucial for the breakdown of the neurotransmitter acetylcholine in the synaptic cleft. By inhibiting this enzyme, Haloxon causes an accumulation of acetylcholine, leading to continuous stimulation of muscles, paralysis, and eventual death of the parasitic worms.
Uses in Veterinary Medicine[edit]
Haloxon is used to treat a variety of parasitic infections in animals, particularly in cattle, sheep, and goats. It is effective against a range of gastrointestinal nematodes and is often administered as part of a broader parasite control program.
Safety and Toxicity[edit]
As with other organophosphates, Haloxon can be toxic to non-target species, including humans, if not used properly. It is important to follow recommended dosages and safety guidelines to minimize the risk of toxicity. Symptoms of organophosphate poisoning include muscle twitching, weakness, respiratory distress, and, in severe cases, death.
Regulation and Approval[edit]
The use of Haloxon is regulated by veterinary health authorities, and it is important to adhere to local regulations regarding its use in livestock. Withdrawal periods must be observed to ensure that residues do not remain in meat or milk intended for human consumption.
Also see[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
