Germabenzene
Germabenzene[edit]
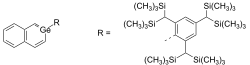
Germabenzene is a chemical compound that belongs to the class of organogermanium compounds. It is a heteroaromatic compound where one of the carbon atoms in the benzene ring is replaced by a germanium atom. This substitution results in unique chemical properties that distinguish germabenzene from its all-carbon counterpart.
Structure and Bonding[edit]
Germabenzene retains the planar, hexagonal structure characteristic of benzene, but the presence of the germanium atom introduces differences in bond lengths and bond angles. The germanium-carbon bonds in germabenzene are typically longer than the carbon-carbon bonds found in benzene due to the larger atomic radius of germanium compared to carbon.
Synthesis[edit]
The synthesis of germabenzene is a complex process that often involves the use of organometallic chemistry techniques. One common method involves the reaction of a germylene precursor with an appropriate aromatic compound under controlled conditions. The synthesis must be carefully managed to prevent the formation of unwanted byproducts and to ensure the stability of the germabenzene product.
Reactivity[edit]
Germabenzene exhibits unique reactivity patterns due to the presence of the germanium atom. It can participate in electrophilic aromatic substitution reactions, similar to benzene, but the reactivity is often enhanced or altered by the germanium atom. This makes germabenzene a valuable compound for studying the effects of heteroatom substitution in aromatic systems.
Applications[edit]
While germabenzene itself is primarily of interest in academic research, its derivatives have potential applications in materials science and organic electronics. The unique electronic properties of germabenzene derivatives make them candidates for use in semiconductors and other advanced materials.
Related Compounds[edit]
Germabenzene is part of a broader class of heteroaromatic compounds that include silabenzene, stannabenzene, and phosphabenzene. These compounds are studied for their unique properties and potential applications in various fields of chemistry and materials science.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian