Functional group
A functional group is a specific group of atoms within a molecule that is responsible for a characteristic of that compound. The same functional group will undergo the same or similar chemical reaction(s) regardless of the rest of the molecule's composition. This concept is central to organic chemistry and is used to predict the properties and reactivity of organic compounds.
Overview[edit]
Functional groups are specific groups of atoms or bonds within molecules that are responsible for the characteristic chemical reactions of those molecules. They are the key structural elements that define the chemical properties of organic compounds. Functional groups are attached to the carbon backbone of organic molecules and determine the types of chemical reactions the molecule can undergo.
Common Functional Groups[edit]
Hydrocarbons[edit]
Hydrocarbons are the simplest organic compounds, consisting entirely of carbon and hydrogen. They can be classified into several types based on the presence of functional groups:
- Alkanes: Saturated hydrocarbons with single bonds only. They are relatively unreactive due to the strength of the C-H and C-C bonds.
- Alkenes: Unsaturated hydrocarbons containing at least one carbon-carbon double bond. The double bond is the functional group that imparts reactivity to alkenes.
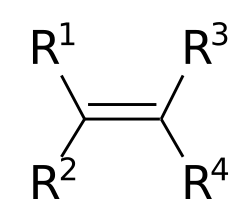
- Alkynes: Unsaturated hydrocarbons with one or more carbon-carbon triple bonds. The triple bond is the functional group that defines alkynes.
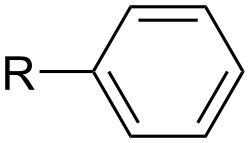
Aromatic Compounds[edit]
Aromatic compounds contain a planar ring of atoms that is stabilized by resonance. The most common example is the benzene ring.
Halides[edit]
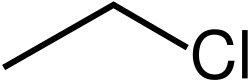
Halides are compounds where a halogen atom (such as chlorine, bromine, or iodine) is bonded to a carbon atom. They are often used as intermediates in organic synthesis.
Alcohols and Ethers[edit]
- Alcohols: Compounds containing a hydroxyl group (-OH) bonded to a carbon atom. They are polar and can form hydrogen bonds, affecting their solubility and boiling points.
- Ethers: Compounds with an oxygen atom connected to two alkyl or aryl groups. They are generally less reactive than alcohols.
Carbonyl Compounds[edit]
Carbonyl compounds contain a carbon-oxygen double bond. This group is highly polar and reactive, making it a key functional group in many organic reactions.
- Aldehydes: Carbonyl group bonded to at least one hydrogen atom.
- Ketones: Carbonyl group bonded to two carbon atoms.
Carboxylic Acids and Derivatives[edit]
- Carboxylic Acids: Contain a carboxyl group (-COOH). They are acidic due to the ability to donate a proton from the hydroxyl group.
- Esters: Derived from carboxylic acids where the hydroxyl group is replaced by an alkoxy group.
Amines and Amides[edit]
- Amines: Contain a nitrogen atom bonded to one or more alkyl or aryl groups. They are basic and can participate in hydrogen bonding.
- Amides: Derived from carboxylic acids where the hydroxyl group is replaced by an amine group.
Importance in Chemistry[edit]
Functional groups are crucial in the study of organic chemistry because they determine the reactivity and properties of molecules. By understanding functional groups, chemists can predict how a molecule will react under certain conditions, design new molecules with desired properties, and synthesize complex organic compounds.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian