Ethylparaben
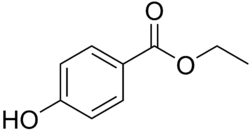

Ethylparaben is a paraben and a paraben ester commonly used as a preservative in cosmetics, pharmaceuticals, and food products. It is the ethyl ester of p-hydroxybenzoic acid. Ethylparaben is known for its antimicrobial properties, which help in extending the shelf life of products by preventing the growth of bacteria, mold, and yeast.
Chemical Properties[edit]
Ethylparaben has the chemical formula C9H10O3 and a molecular weight of 166.17 g/mol. It appears as a white crystalline powder that is soluble in alcohol and slightly soluble in water. The compound is synthesized through the esterification of p-hydroxybenzoic acid with ethanol.
Uses[edit]
Ethylparaben is widely used in various industries due to its effective preservative qualities:
- In cosmetics, it is used in products such as shampoos, lotions, and makeup to prevent microbial contamination.
- In pharmaceuticals, it is used in creams, ointments, and liquid medications to ensure product stability and safety.
- In food products, it is used as a preservative to extend shelf life and maintain product quality.
Safety and Regulation[edit]
Ethylparaben, like other parabens, has been the subject of safety evaluations. Regulatory agencies such as the Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have deemed ethylparaben safe for use in specified concentrations. However, there has been ongoing debate and research regarding the potential health effects of parabens, particularly their role as endocrine disruptors.
Related Compounds[edit]
Ethylparaben is part of a larger group of paraben esters, which include:
These compounds share similar preservative properties and are often used in combination to enhance their antimicrobial efficacy.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
