Electron
Subatomic particle with negative electric charge
Electron[edit]
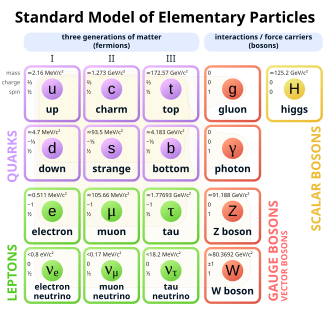
The electron is a subatomic particle with a negative electric charge. It is one of the fundamental constituents of matter, playing a crucial role in electricity, magnetism, and chemical bonding. Electrons are part of the lepton family of particles and are considered elementary particles because they are not composed of smaller particles.
History[edit]
The concept of the electron emerged from studies of electricity in the 19th century. The term "electron" was coined by George Johnstone Stoney in 1891, but it was J.J. Thomson who is credited with the discovery of the electron in 1897 through his experiments with cathode rays.

Properties[edit]
Electrons have a rest mass of approximately 9.109 × 10−31 kilograms, which is about 1/1836 of the mass of a proton. They carry a negative electric charge of approximately −1.602 × 10−19 coulombs. Electrons exhibit both particle and wave properties, a duality that is a fundamental aspect of quantum mechanics.

Quantum Mechanics[edit]
In the realm of quantum mechanics, electrons are described by wave functions and can exist in various quantum states. The behavior of electrons in atoms is described by atomic orbitals, which are regions of space where there is a high probability of finding an electron.


Role in Atoms[edit]
Electrons are bound to the nucleus of an atom by the electromagnetic force. They occupy energy levels or shells around the nucleus, and their arrangement determines the chemical properties of an element. The Bohr model of the atom, proposed by Niels Bohr, was an early attempt to describe electron behavior in atoms.

Interactions[edit]
Electrons interact with other particles through the electromagnetic force. They can also emit or absorb photons, which are quanta of light, during transitions between energy levels. In addition, electrons can form virtual particles in the quantum field theory framework.

Applications[edit]
Electrons are fundamental to the operation of many modern technologies, including semiconductors, transistors, and cathode ray tubes. They are also essential in the study of electricity and magnetism, as they are the primary charge carriers in conductors.

Lorentz Force[edit]
The motion of electrons in a magnetic field is described by the Lorentz force, which is the force experienced by a charged particle moving through a magnetic field.

Related pages[edit]
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian