Neutron

A neutron is a subatomic particle that is found in the atomic nucleus of atoms, along with protons. Neutrons have no net electric charge and a mass slightly greater than that of protons. They are a key component of atomic nuclei and play a crucial role in the stability and properties of atoms.
Structure and Properties[edit]
Neutrons are composed of three quarks: two down quarks and one up quark, held together by the strong nuclear force mediated by gluons. The absence of electric charge in neutrons makes them electrically neutral, which allows them to penetrate atomic nuclei without being repelled by the positive charge of protons.
The mass of a neutron is approximately 1.675 × 10-27 kg, which is slightly greater than that of a proton. Neutrons are unstable when isolated and undergo beta decay with a half-life of about 14 minutes, transforming into a proton, an electron, and an antineutrino.
Role in Nuclear Reactions[edit]
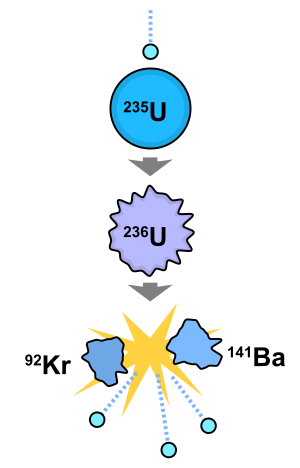
Neutrons are essential in nuclear reactions, including nuclear fission and nuclear fusion. In fission, a neutron collides with a heavy nucleus, causing it to split into smaller nuclei and release additional neutrons and energy. This process is the basis for nuclear power and atomic bombs.
In fusion, light nuclei combine to form a heavier nucleus, releasing energy. Neutrons are often a byproduct of fusion reactions, such as those occurring in the sun and other stars.
Neutron Decay[edit]

Neutrons undergo beta decay, a type of radioactive decay where a neutron is converted into a proton, an electron, and an antineutrino. This process is crucial in the transformation of elements and isotopes in nature and is a key mechanism in the decay of radioactive materials.
Applications[edit]
Neutrons have numerous applications in science and technology. They are used in neutron scattering experiments to study the structure of materials at the atomic level. Neutron imaging is a technique used to visualize the internal structure of objects, similar to X-ray imaging but with different contrast properties.

Neutron Sources[edit]
Neutrons are produced in various ways, including nuclear reactors, particle accelerators, and radioactive decay. Nuclear reactors are a primary source of neutrons for research and industrial applications. The Institut Laue–Langevin in France is one of the leading facilities for neutron research.

Related Pages[edit]
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends ✔ Tele visits available with certain limitations Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
- Budget GLP-1 shots
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian