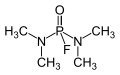
Dimefox
Dimefox is a highly toxic organophosphate compound that is used as an insecticide. It is a clear, colorless liquid that is soluble in water and most organic solvents. Dimefox is a potent acetylcholinesterase inhibitor, which means it can disrupt the functioning of the nervous system in both insects and mammals.
Chemical Properties[edit]
Dimefox has the chemical formula C6H12N2O3PS. It is a derivative of phosphoric acid, and is classified as an organophosphate due to the presence of a phosphorus atom bonded to an oxygen atom and a carbon atom in its molecular structure. The compound is highly reactive, and can undergo hydrolysis to form a variety of breakdown products.
Toxicity[edit]
Dimefox is highly toxic to both insects and mammals. It works by inhibiting the enzyme acetylcholinesterase, which is essential for the proper functioning of the nervous system. This inhibition leads to an accumulation of acetylcholine, a neurotransmitter, in the synapses of the nervous system. The resulting overstimulation of the nervous system can lead to a variety of symptoms, including muscle weakness, convulsions, and in severe cases, death.
Use in Pest Control[edit]
Despite its high toxicity, Dimefox is used as an insecticide due to its effectiveness in controlling a wide range of insect pests. It is particularly effective against coleopteran insects, such as beetles and weevils, as well as against lepidopteran insects, such as moths and butterflies. However, due to its high toxicity to mammals, its use is strictly regulated in many countries.
Safety and Regulation[edit]
Due to its high toxicity, the use and handling of Dimefox is subject to strict regulation in many countries. In the United States, for example, the Environmental Protection Agency (EPA) classifies Dimefox as a Restricted Use Pesticide (RUP), which means it can only be used by certified applicators. In addition, the EPA has set strict limits on the amount of Dimefox that can be released into the environment.
See Also[edit]
-
3D model of Dimefox
-
Chemical structure of Dimefox
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian