Diketene
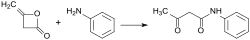
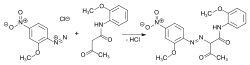
Diketene is a chemical compound with the formula C_4H_4O_2. It is a member of the ketene family and is characterized by its unique structure, which includes two ketone groups (hence the prefix "di-") linked by a ethylene bridge. Diketene is a highly reactive, colorless liquid at room temperature and is used in the synthesis of various acetoacetic acid derivatives, acetic anhydride, and other chemicals.
Properties and Structure[edit]
Diketene is an unsaturated dimer of ketene and exhibits both electrophilic and nucleophilic properties due to its reactive carbonyl groups. The molecule adopts a strained, four-membered ring structure, which contributes to its high reactivity. It is soluble in common organic solvents but hydrolyzes in the presence of water to form acetic acid and acetoacetate.
Synthesis[edit]
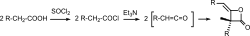
Diketene is typically synthesized through the dimerization of ketene, which itself is produced via the dehydrative pyrolysis of acetic acid or the dehydration of ethanol. The process requires precise control of temperature and pressure to favor the formation of diketene over other by-products.
Applications[edit]
Diketene's reactivity makes it a valuable intermediate in organic synthesis. It is used in the production of:
- Acetoacetate esters, which are precursors to dyes, pigments, and pharmaceuticals.
- Amides and esters of acetoacetic acid, used in the manufacture of antibiotics and other medicinal compounds.
- Acetic anhydride, an important reagent in organic chemistry and the production of cellulose acetate.
Safety and Handling[edit]
Diketene is a hazardous material that requires careful handling. It is corrosive and can cause severe burns upon contact with skin or eyes. Inhalation of diketene vapors can lead to respiratory irritation and long-term health effects. Appropriate safety measures, including the use of personal protective equipment (PPE) and proper ventilation, are essential when working with diketene.
Environmental Impact[edit]
The production and use of diketene must be managed to minimize environmental impact. Its reactivity and potential to hydrolyze can lead to the release of organic acids into the environment, necessitating careful waste management and disposal practices.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
