Diisopropylamine
Diisopropylamine[edit]
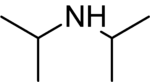
Diisopropylamine is a secondary amine with the chemical formula (CH_)_CHNHCH(CH_)_. It is a colorless liquid with a strong ammonia-like odor. Diisopropylamine is commonly used as a precursor to other chemical compounds and as a base in organic synthesis.
Chemical Properties[edit]
Diisopropylamine is a secondary amine, meaning it has two alkyl groups attached to the nitrogen atom. This structure contributes to its basicity and reactivity. The presence of the isopropyl groups makes it more sterically hindered compared to primary amines, affecting its reactivity in certain chemical reactions.
Synthesis[edit]
Diisopropylamine can be synthesized through the alkylation of ammonia with isopropyl alcohol in the presence of a catalyst. Another method involves the reaction of isopropyl chloride with ammonia under controlled conditions.
Applications[edit]
Diisopropylamine is widely used in the production of lithium diisopropylamide (LDA), a strong non-nucleophilic base used in organic synthesis. LDA is particularly useful for deprotonating weakly acidic compounds, allowing for the formation of carbanions that can participate in further chemical reactions.
Safety and Handling[edit]
Diisopropylamine is a flammable liquid and should be handled with care. It can cause irritation to the skin, eyes, and respiratory system. Proper personal protective equipment (PPE) such as gloves and goggles should be worn when handling this chemical.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian