Dicarbonyl
Dicarbonyl compounds are a group of organic compounds characterized by the presence of two carbonyl groups (C=O) within their molecular structure. These compounds play a significant role in various biochemical and industrial processes. The presence of two carbonyl groups often imparts unique chemical properties, making dicarbonyls important intermediates in organic synthesis, as well as in the formation of advanced glycation end-products (AGEs), which are relevant in medical and nutritional sciences.
Structure and Classification[edit]
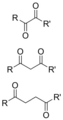
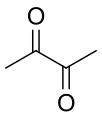
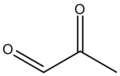
Dicarbonyl compounds can be classified based on the positioning of their carbonyl groups. If the carbonyl groups are adjacent to each other, the compound is referred to as a 1,2-dicarbonyl compound. Alternatively, if there is one carbon atom separating the carbonyl groups, the compound is known as a 1,3-dicarbonyl compound. There are also 1,4-dicarbonyl compounds, where two carbon atoms separate the carbonyl groups.
Chemical Properties[edit]
The chemical reactivity of dicarbonyl compounds is largely influenced by the electron-withdrawing effect of the carbonyl groups. This effect can lead to increased acidity of the hydrogen atoms adjacent to the carbonyl groups, making dicarbonyls susceptible to nucleophilic attacks. This property is exploited in various organic synthesis reactions, such as the Michael Addition and the Aldol Condensation.
Biological Significance[edit]
In biological systems, dicarbonyl compounds are formed as intermediates in the metabolism of sugars and fats. One of the most studied dicarbonyls in biology is Methylglyoxal, a highly reactive compound that can react with proteins, nucleic acids, and other macromolecules to form AGEs. The accumulation of AGEs is associated with aging and various diseases, including diabetes, cardiovascular diseases, and neurodegenerative disorders.
Industrial Applications[edit]
Dicarbonyl compounds are utilized in the synthesis of a wide range of industrial products, including pharmaceuticals, agrochemicals, and polymers. Their reactivity makes them valuable building blocks in organic synthesis, enabling the construction of complex molecular architectures.
Health Implications[edit]
The formation and accumulation of dicarbonyl compounds, particularly AGEs, in the human body have been linked to several health issues. Dietary intake of foods high in dicarbonyls or their precursors can contribute to the body's AGE pool, exacerbating conditions related to oxidative stress and inflammation. Research into inhibitors of dicarbonyl compound formation and scavengers of these reactive species is ongoing, with the aim of mitigating their harmful effects.
See Also[edit]
-
Dicarbonyl structure
-
Diacetyl
-
Methylglyoxal
-
Ketocarboxylic Acids General Formulae
-
Acetylacetone
-
p-Benzoquinone
-
Paal-Knorr Pyrrole Synthesis
-
Succinaldehyde Hydrate
-
Orthophthalaldehyde and hydrated forms
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian