Chloroprene

Chloroprene is an organic compound with the formula CH2=CCl−CH=CH2. It is a colorless liquid that is used primarily in the production of neoprene, a type of synthetic rubber. Neoprene is known for its various desirable properties, including good chemical stability and resistance to heat, chemicals, and physical wear, making it useful in a wide range of applications from automotive and marine parts to electrical insulation and wetsuits.
Production[edit]
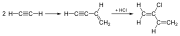
Chloroprene is manufactured through the chlorination of butadiene, a process that can be conducted in a few different ways, including acetylene and butylene methods. The most common method involves the chlorination of butadiene to produce 3,4-dichloro-1-butene, which is then dehydrochlorinated to produce chloroprene.
Properties and Uses[edit]
Chloroprene is notable for its ability to polymerize. The polymerization can be initiated by free radicals, and this process is used to produce neoprene rubber. Neoprene exhibits excellent physical and chemical properties, such as resistance to oil, chemicals, and weathering, making it suitable for industrial applications including hoses, belts, gaskets, and O-rings. It is also used in consumer products like gloves, waders, and orthopedic braces, offering comfort, support, and durability.
Health and Safety[edit]
Exposure to chloroprene can pose health risks, including irritation of the eyes, skin, and respiratory system. Long-term exposure has been associated with more serious health effects, such as liver damage and cancer. As a result, workplace exposure limits have been established in many countries to protect workers in industries that manufacture or use chloroprene.
Environmental Impact[edit]
Chloroprene is volatile and can be released into the air from industrial sites, leading to concerns about its impact on air quality and public health. Environmental agencies monitor and regulate its emission to minimize its release into the environment. Efforts to reduce chloroprene emissions include the use of control technologies at manufacturing plants and ongoing research into alternative materials with lower environmental impacts.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
