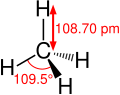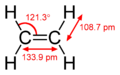Carbon–hydrogen bond
Carbon–hydrogen bond (C–H bond) is a chemical bond that exists between carbon atoms and hydrogen atoms in organic compounds. This bond is a type of covalent bond, where two atoms share a pair of electrons. The C–H bond is one of the most fundamental and widespread bonds in organic chemistry and biochemistry, playing a crucial role in the structure and reactivity of organic molecules.
Characteristics[edit]
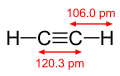
The carbon–hydrogen bond is characterized by its bond length, bond energy, and its role in the molecular structure. The typical bond length of a C–H bond is approximately 1.09 Å (angstroms), and its bond energy varies depending on the hybridization of the carbon atom; it is about 413 kJ/mol for sp3 hybridized carbon atoms, 423 kJ/mol for sp2 hybridized carbon atoms, and 439 kJ/mol for sp hybridized carbon atoms.
Types of C–H Bonds[edit]
Depending on the hybridization state of the carbon atom to which the hydrogen is bonded, C–H bonds can be classified into three main types:
- Aliphatic C–H bonds: These are found in alkanes, alkenes, and alkynes, where the carbon atoms are sp3, sp2, and sp hybridized, respectively.
- Aromatic C–H bonds: These occur in aromatic compounds, such as benzene, where the carbon atoms are part of an aromatic ring and are sp2 hybridized.
- Vinylic and allylic C–H bonds: These are present in vinyl (sp2 hybridized carbon adjacent to a double bond) and allyl (sp3 hybridized carbon adjacent to a double bond) groups, respectively.
Reactivity[edit]
The reactivity of the C–H bond is a central theme in organic synthesis and biochemistry. Although C–H bonds are relatively inert under standard conditions, their activation can lead to a wide range of chemical transformations. Techniques for C–H bond activation include the use of catalysts, such as transition metals, and the application of light or heat.
Importance in Organic Synthesis[edit]
In organic synthesis, the functionalization of C–H bonds is a powerful strategy for constructing complex molecules from simpler ones. This approach can streamline synthetic routes and increase the efficiency of chemical synthesis.
Biological Significance[edit]
In biochemistry, the C–H bond is found in all organic compounds that make up living organisms, including carbohydrates, lipids, proteins, and nucleic acids. Enzymes, such as cytochrome P450s, can selectively activate C–H bonds in complex molecules, facilitating a wide range of biochemical transformations.
Environmental Impact[edit]
The combustion of hydrocarbons, which involves the breaking of C–H bonds, is a major source of energy in the modern world. However, this process releases carbon dioxide, a greenhouse gas, contributing to global warming and climate change.
See Also[edit]
-
Methane molecule with dimensions
-
Ethane molecule in staggered conformation with dimensions
-
Ethylene molecule with dimensions
-
Acetylene molecule with dimensions
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian