Carbodiimide
Carbodiimide is a class of organic compounds characterized by the presence of the functional group N=C=N. This group consists of a carbon atom double-bonded to two nitrogen atoms. Carbodiimides are widely used in organic synthesis and materials science for the formation of amide bonds, making them valuable reagents in the production of pharmaceuticals, polymers, and other chemical products.
Chemistry[edit]
Carbodiimides are formed through the dehydration of urea or from thiourea. The general chemical formula for carbodiimides is R-N=C=N-R', where R and R' can be the same or different alkyl or aryl groups. They are reactive towards nucleophiles, including water, alcohols, and amines, making them useful for a variety of chemical reactions.
Synthesis[edit]
The synthesis of carbodiimides typically involves the dehydration of urea derivatives or the reaction of phosgene with primary amines. One of the most common methods for preparing carbodiimides is the dehydration of urea or thiourea in the presence of a dehydrating agent, such as phosphorus oxychloride.
Reactivity[edit]
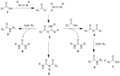
Carbodiimides are best known for their role in the formation of amide bonds from carboxylic acids and amines, a process known as amidation. This reaction is crucial in peptide synthesis, where carbodiimides, such as dicyclohexylcarbodiimide (DCC) and diisopropylcarbodiimide (DIC), are used to couple amino acids. They can also be used to activate phosphate groups in the synthesis of nucleotides and DNA.
Applications[edit]
Carbodiimides find applications in various fields, including:
- Pharmaceuticals: In drug synthesis, especially in peptide coupling reactions.
- Materials Science: In the preparation of polymers and coatings, where they are used to cross-link polymers.
- Bioconjugation: For attaching biomolecules to one another or to solid supports, crucial in the development of biosensors and diagnostic tools.
Safety and Handling[edit]
Carbodiimides are generally handled with care due to their reactivity. They can cause skin and respiratory irritation, and their reactions can release toxic by-products. Proper safety measures, including the use of personal protective equipment (PPE) and working in a fume hood, are recommended when handling these compounds.
Environmental Impact[edit]
The environmental impact of carbodiimides depends on their specific applications and disposal methods. As with many chemical reagents, responsible handling, usage, and disposal practices are essential to minimize any negative environmental effects.
Carbodiimide[edit]
-
General structure of carbodiimides
-
Crystal structure of a carbodiimide compound
-
Carbodiimide-mediated amide coupling scheme
-
Mechanism of carbodiimide reaction
-
Structure of DCC (Dicyclohexylcarbodiimide)
-
Structure of DIC (Diisopropylcarbodiimide)