Butyl group
Butyl group is a four-carbon substituent in organic chemistry. It is derived from the alkane butane and has the molecular formula C4H9. The term butyl is used in organic chemistry to describe the butyl functional group. The prefix "butyl" is derived from the word "butyric," which in turn comes from the Latin "butyrum," meaning butter.
Structure and properties[edit]
The butyl group, with four carbon atoms connected to each other in a straight chain, is a type of alkyl group. It is a part of larger molecules and can be found in various types of compounds, including alcohols, ethers, and halides. The butyl group is nonpolar and hydrophobic, meaning it does not mix well with water. However, it can dissolve in organic solvents.
Types of butyl groups[edit]
There are four isomers of the butyl group, each with a different structure:
- n-Butyl group (n-C4H9): This is the unbranched isomer with the structure CH3CH2CH2CH2.
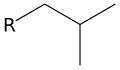
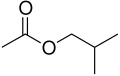
- Isobutyl group (i-C4H9): This is the isomer with the structure (CH3)2CHCH2.
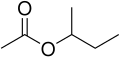
- Sec-butyl group (s-C4H9): This is the isomer with the structure CH3CH2CH(CH3).
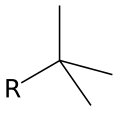
- Tert-butyl group (t-C4H9): This is the highly branched isomer with the structure (CH3)3C.
Uses[edit]
Butyl groups are commonly used in organic synthesis. They can be added to other molecules to increase their size or to change their properties. For example, butyl groups are often used to increase the hydrophobicity of a molecule, making it more soluble in organic solvents.
See also[edit]
References[edit]
-
Sec-butyl acetate
-
N-Butyl group
-
Sec-Butyl group
-
Isobutyl group
-
Tert-Butyl group
-
Butyl acetate
-
Isobutyl acetate
-
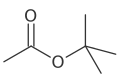
Tert-Butyl acetate
-
Tert-butyl effect
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian