Bisphenol AF
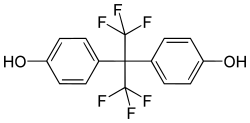
Bisphenol AF (BPAF) is a chemical compound that belongs to the family of bisphenols. It is used in the production of various polymers and plastics, and is a substitute for Bisphenol A (BPA) in some applications.
Chemical Structure and Properties[edit]
Bisphenol AF is an organic compound with the chemical formula C20H14F4O2. It is a white crystalline powder at room temperature. The compound consists of two phenol groups linked by a carbon atom, with two fluorine atoms attached to each phenol group. This structure gives BPAF its unique properties, including high thermal stability and resistance to acids and bases.
Production and Uses[edit]
Bisphenol AF is synthesized through the reaction of phenol with hexafluoroacetone in the presence of an acid catalyst. The resulting compound is then purified and processed into its final form.
BPAF is used in the production of various types of plastics and polymers, including polyesters, polycarbonates, and epoxy resins. These materials are used in a wide range of applications, from packaging materials and construction materials to electronics and automotive parts.
Health and Environmental Concerns[edit]
Like other bisphenols, Bisphenol AF has been the subject of health and environmental concerns. Studies have suggested that BPAF may have endocrine disrupting properties, potentially interfering with the body's hormone system. There is also concern about the persistence of BPAF in the environment and its potential effects on wildlife.
Regulation[edit]
In many countries, the use of Bisphenol AF is regulated due to its potential health and environmental impacts. Regulations may include restrictions on its use in certain applications, requirements for its safe handling and disposal, and mandates for its labeling on products.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
