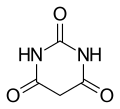
Barbituric acid
Barbituric acid is an organic compound that is the parent structure for a variety of barbiturates, which are widely used as sedative and anesthetic drugs. The structure of barbituric acid is based on a pyrimidine ring, which is a six-membered ring with two nitrogen atoms and four carbon atoms. The name "barbituric acid" comes from the fact that it was first synthesized on Saint Barbara's Day in 1864 by German chemist Adolf von Baeyer.
History[edit]
Barbituric acid was first synthesized on December 4, 1864, by Adolf von Baeyer. This was done by condensing urea (an animal waste product) with diethyl malonate (an ester derived from the natural substance malonic acid). The reaction created a substance that was crystalline, slightly acidic, and was named barbituric acid in honor of Saint Barbara, the patron saint of architecture and fireworks, as well as the name of Baeyer's friend Barbara.
Structure and properties[edit]
Barbituric acid is a six-membered heterocyclic compound with two nitrogen atoms and four carbon atoms in the ring. It is a white crystalline substance that is slightly soluble in water and freely soluble in hot water. It is also soluble in alcohol and slightly soluble in ether.
The structure of barbituric acid is the basis for the structure of barbiturates, a class of drugs that act as central nervous system depressants. Barbiturates are derived from barbituric acid by substitution of one or more of the hydrogen atoms by alkyl, alkenyl or aryl groups.
Synthesis[edit]
Barbituric acid can be synthesized by the reaction of malonic acid with urea in the presence of phosphorus oxychloride (POCl3) as a catalyst. The reaction proceeds via a condensation reaction, with the elimination of a molecule of water.
Uses[edit]
While barbituric acid itself is not used clinically, its derivatives, known as barbiturates, have been widely used as sedative and anesthetic drugs. Barbiturates have largely been replaced by safer drugs, although some are still in use for specific purposes.
See also[edit]
Barbituric acid gallery[edit]
-
Barbituric acid structural formula
-
Barbituric acid 3D ball model
-
Barbituric acid synthesis
-
Barbituric acid, acid reaction
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian