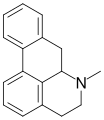
Aporphine
Aporphine alkaloids and their pharmacological significance
Aporphine is a class of alkaloids that are structurally related to the benzylisoquinoline alkaloids. These compounds are characterized by a tetracyclic structure that includes a phenanthrene core. Aporphine alkaloids are found in various plant species and have been studied for their diverse pharmacological properties.
Structure and Chemistry[edit]
Aporphine alkaloids possess a unique tetracyclic structure that is derived from the benzylisoquinoline skeleton. The core structure consists of a phenanthrene ring system, which is a tricyclic aromatic hydrocarbon, fused with a nitrogen-containing heterocycle. This configuration is responsible for the diverse biological activities exhibited by these compounds.
Sources[edit]
Aporphine alkaloids are primarily found in the Magnoliaceae and Menispermaceae families. Notable plants containing aporphine alkaloids include species of the genera Nymphaea, Annona, and Stephania. These plants have been used in traditional medicine for their therapeutic properties.
Pharmacological Properties[edit]
Aporphine alkaloids exhibit a wide range of pharmacological activities, including:
- Dopaminergic Activity: Some aporphine derivatives act as dopamine receptor agonists or antagonists, influencing neurological pathways and potentially offering therapeutic benefits in conditions such as Parkinson's disease.
- Antioxidant Activity: Certain aporphine compounds have been shown to possess antioxidant properties, which may protect cells from oxidative stress and reduce the risk of chronic diseases.
- Antimicrobial Activity: Aporphine alkaloids have demonstrated antimicrobial effects against a variety of bacteria and fungi, suggesting potential applications in treating infections.
- Anticancer Activity: Research has indicated that some aporphine derivatives may inhibit the growth of cancer cells, making them candidates for further investigation in cancer therapy.
Biosynthesis[edit]
The biosynthesis of aporphine alkaloids involves the enzymatic conversion of tyrosine to dopamine, which then undergoes a series of transformations to form the benzylisoquinoline precursor. This precursor is further modified through oxidative coupling and rearrangement to produce the aporphine structure.
Applications in Medicine[edit]
Due to their diverse biological activities, aporphine alkaloids are of interest in the development of new therapeutic agents. Their potential applications include:
- Treatment of neurological disorders such as Parkinson's disease.
- Development of new antimicrobial agents to combat resistant strains of bacteria and fungi.
- Exploration as anticancer agents due to their ability to inhibit tumor growth.
Related Pages[edit]
Aporphine gallery[edit]
-
Aporphine
-
Aporphines
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian