Aminomethanol
Aminomethanol (also known as Methanolamine) is a chemical compound with the formula CH3NOH. It is the simplest form of amino alcohols, which are compounds containing both an amino group (-NH2) and a hydroxyl group (-OH). Aminomethanol is a key building block in the chemical industry and is used in the synthesis of a wide range of products, including pharmaceuticals, agrochemicals, and resins.
Structure and Properties[edit]
Aminomethanol consists of a methanol molecule in which one of the hydrogen atoms is replaced by an amino group. This modification imparts unique properties to the molecule, making it both a weak base and a weak acid. It is a colorless, hygroscopic liquid at room temperature and has a high boiling point relative to its molecular weight due to the presence of both amino and hydroxyl groups, which facilitate hydrogen bonding.
Synthesis[edit]
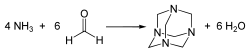
Aminomethanol can be synthesized through several methods. One common approach is the reaction of ammonia with ethylene oxide, followed by hydrolysis of the intermediate to produce aminomethanol. Another method involves the direct amination of methanol under high pressure and temperature in the presence of a catalyst.
Applications[edit]
Due to its dual functionality, aminomethanol is a versatile intermediate in organic synthesis. It is used in the manufacture of herbicides, detergents, and varnishes. In the pharmaceutical industry, it serves as a precursor to certain drugs and is involved in the production of vitamins and antibiotics. Additionally, aminomethanol is a component in the formulation of cosmetics and textile finishing agents.
Safety and Environmental Impact[edit]
Aminomethanol is considered to be of moderate toxicity. It can cause irritation to the skin, eyes, and respiratory tract upon exposure. Proper handling and storage procedures are essential to minimize health risks. In the environment, aminomethanol can be broken down by microorganisms, reducing its potential for long-term environmental impact. However, its release into water bodies should be avoided as it can pose risks to aquatic life.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
