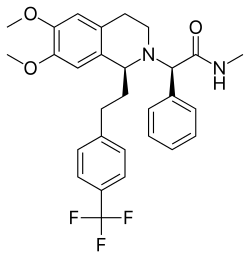
Almorexant
Overview[edit]
Almorexant is a drug that was developed as a dual orexin receptor antagonist (DORA) for the treatment of insomnia. Orexins are neuropeptides that play a crucial role in regulating the sleep-wake cycle and arousal. By blocking the action of orexins, almorexant promotes sleep.
Mechanism of Action[edit]
Almorexant works by inhibiting the binding of orexins to their receptors, specifically orexin receptor 1 (OX1R) and orexin receptor 2 (OX2R). This inhibition reduces the wake-promoting effects of orexins, thereby facilitating the onset and maintenance of sleep.
Development and Clinical Trials[edit]
Almorexant was developed by Actelion Pharmaceuticals in collaboration with GlaxoSmithKline. It underwent several clinical trials to assess its efficacy and safety in treating insomnia. However, despite initial promising results, the development of almorexant was discontinued due to concerns about its safety profile, including potential liver toxicity.
Pharmacokinetics[edit]
The pharmacokinetic profile of almorexant includes its absorption, distribution, metabolism, and excretion. Almorexant is administered orally and is metabolized primarily in the liver. The drug's half-life and bioavailability were key considerations during its development.
Side Effects[edit]
Common side effects observed in clinical trials of almorexant included drowsiness, headache, and dizziness. More serious adverse effects, such as liver enzyme elevation, led to the discontinuation of its development.
Potential Applications[edit]
While almorexant itself is no longer being developed, the concept of orexin receptor antagonism remains a promising avenue for the treatment of insomnia and other sleep disorders. Other DORAs are currently being investigated for their therapeutic potential.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian