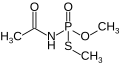
Acephate
An organophosphate insecticide
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
Acephate is an organophosphate insecticide used in agriculture to control a wide range of insect pests. It is known for its systemic action, meaning it can be absorbed by plants and transported throughout their tissues, providing protection against insects that feed on the plant.
Chemical properties[edit]
Acephate is a white solid that is soluble in water. It is chemically classified as an organophosphate, which means it contains phosphorus as part of its molecular structure. The chemical formula for acephate is C_H__NO_PS.
Mode of action[edit]
Acephate works by inhibiting the enzyme acetylcholinesterase, which is essential for the proper functioning of the nervous system in insects. By disrupting this enzyme, acephate causes an accumulation of acetylcholine, leading to paralysis and death of the insect.
Uses[edit]
Acephate is used on a variety of crops, including cotton, soybeans, tobacco, and vegetables. It is effective against a range of pests such as aphids, thrips, and caterpillars.
Safety and environmental impact[edit]
As an organophosphate, acephate poses potential risks to human health and the environment. It is important to follow safety guidelines when handling and applying acephate to minimize exposure. The use of personal protective equipment (PPE) is recommended for individuals applying this insecticide.
Regulation[edit]
Acephate is regulated by various governmental agencies to ensure its safe use. In the United States, the Environmental Protection Agency (EPA) oversees the registration and use of acephate, setting limits on its application to protect human health and the environment.
Related pages[edit]
Gallery[edit]
-
Structural formula of Acephate
-
3D model of Acephate
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian