4-Ethylphenol
A phenolic compound found in wine and other fermented products
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
4-Ethylphenol is a phenolic compound with the chemical formula C8H10O. It is a derivative of phenol and is characterized by an ethyl group attached to the para position of the phenol ring.
Occurrence[edit]
4-Ethylphenol is commonly found in wine, where it is produced by the action of certain yeast and bacteria during the fermentation process. It is also present in other fermented products such as beer and cider.
Production[edit]
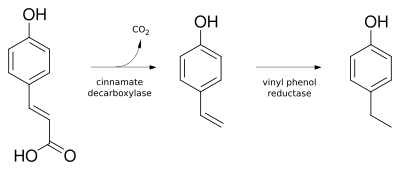
4-Ethylphenol can be synthesized from coumaric acid through a decarboxylation reaction. This process is often facilitated by microbial activity, particularly by species of the genus Brettanomyces.
Properties[edit]
4-Ethylphenol is a colorless liquid with a characteristic phenolic odor. It is slightly soluble in water but more soluble in organic solvents. The presence of 4-ethylphenol in wine is often associated with off-flavors described as "barnyard" or "medicinal".
Applications[edit]
While 4-ethylphenol is generally considered an undesirable compound in wine, it has applications in the synthesis of other chemical compounds. It can be used as an intermediate in the production of pharmaceuticals and fragrances.
Health Effects[edit]
Exposure to 4-ethylphenol can occur through the consumption of contaminated beverages. In small amounts, it is not considered harmful, but excessive exposure may lead to adverse health effects. It is important for winemakers to monitor and control the levels of 4-ethylphenol in their products.
Related pages[edit]
-
P-Ethylphenol
-
Coumaric acid to 4-ethylphenol
-
Whiplash
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian