4-Dimethylaminophenol
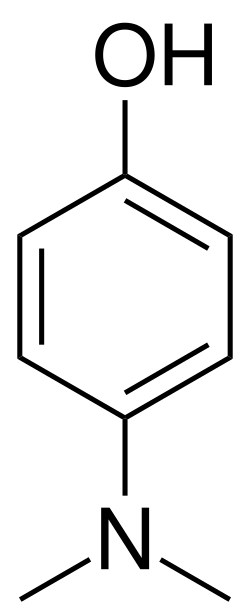
4-Dimethylaminophenol (4-DMAP) is a chemical compound used in the treatment of cyanide poisoning. It is an aromatic amine and a derivative of phenol. The compound is known for its ability to induce the formation of methemoglobin, which can bind cyanide ions and thus detoxify them.
Chemical Structure and Properties[edit]
4-Dimethylaminophenol has the chemical formula C8H11NO and a molecular weight of 137.18 g/mol. It consists of a benzene ring substituted with a hydroxyl group (-OH) and a dimethylamino group (-N(CH3)2) at the para position.
Mechanism of Action[edit]
The primary mechanism of action of 4-Dimethylaminophenol in cyanide poisoning involves the induction of methemoglobin formation. Methemoglobin is a form of hemoglobin in which the iron in the heme group is in the ferric (Fe3+) state, rather than the ferrous (Fe2+) state. Methemoglobin has a high affinity for cyanide ions, which allows it to sequester cyanide from cytochrome c oxidase, an enzyme critical for cellular respiration. This sequestration helps to mitigate the toxic effects of cyanide on the body.
Medical Use[edit]
4-Dimethylaminophenol is administered intravenously in cases of acute cyanide poisoning. It is often used in conjunction with other antidotes such as sodium thiosulfate and hydroxocobalamin. The combination of these agents provides a multi-faceted approach to detoxifying cyanide and restoring normal cellular respiration.
Side Effects and Precautions[edit]
The administration of 4-Dimethylaminophenol can lead to the formation of methemoglobinemia, a condition where an excessive amount of methemoglobin is present in the blood, reducing its oxygen-carrying capacity. Symptoms of methemoglobinemia include cyanosis, headache, dizziness, and shortness of breath. Therefore, the use of 4-Dimethylaminophenol must be carefully monitored, and methemoglobin levels should be regularly checked during treatment.
Synthesis[edit]
The synthesis of 4-Dimethylaminophenol typically involves the nitration of phenol to produce 4-nitrophenol, followed by reduction to 4-aminophenol. The final step involves the methylation of the amino group to yield 4-Dimethylaminophenol.
Related Compounds[edit]
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian