2-Phenylphenol
Chemical compound
| 2-Phenylphenol | |
|---|---|
| 2-Phenylphenol.svg | |
| Identifiers | |
| CAS Number | |
| PubChem CID | 7017 |
| ChemSpider ID | |
| UNII | 8E4OP7R1RK |
| ChEBI | 5153 |
| ChEMBL | 12345 |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | White solid |
| Density | 1.25 g/cm³ |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
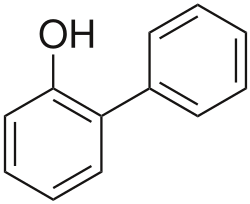
2-Phenylphenol (also known as o-Phenylphenol or Biphenyl-2-ol) is an organic compound with the formula C12H10O. It is a white solid that is slightly soluble in water but more soluble in organic solvents.
Chemical Properties[edit]
2-Phenylphenol is a biphenyl derivative where one of the phenyl groups is substituted with a hydroxyl group at the ortho position. It has a melting point of 57°C and a boiling point of 280°C. The compound has a density of 1.25 g/cm³ and a refractive index of 1.597.
Uses[edit]
2-Phenylphenol is primarily used as a fungicide and bactericide. It is commonly applied to the surface of citrus fruits to prevent mold growth during storage and transportation. Additionally, it is used in the formulation of disinfectants and preservatives for various products, including cosmetics, leather, and textiles.
Safety and Toxicology[edit]
2-Phenylphenol has a flash point of 135°C and an autoignition temperature of 580°C. The LD50 (lethal dose for 50% of the population) for oral intake in rats is 2730 mg/kg, indicating moderate toxicity. It is classified with the GHS signal word "Warning" due to its potential health hazards.
Related Compounds[edit]
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian