2-Pentanol
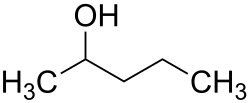
2-Pentanol (IUPAC name: pentan-2-ol), also known by its systematic name sec-pentyl alcohol, is an organic compound with the formula C5H12O. It is a colorless liquid with a slightly unpleasant odor, which is considered a secondary alcohol due to the presence of the hydroxyl group (-OH) attached to the second carbon of the pentane chain. 2-Pentanol is one of the isomers of pentanol.
Properties[edit]
2-Pentanol has a molecular weight of 88.15 g/mol and boils at 119°C. It is moderately soluble in water and miscible with most organic solvents, such as ethanol, diethyl ether, and chloroform. The presence of the hydroxyl group makes it capable of forming hydrogen bonds, which significantly influences its physical properties, such as boiling point and solubility.
Production[edit]
2-Pentanol can be produced through several methods, including the hydrolysis of 2-pentyl halides or the hydrogenation of pentanal. Another common method is through the hydration of pentene, a process that can occur in the presence of acid or base catalysts.
Uses[edit]
2-Pentanol is used in the manufacture of other chemicals, serving as an intermediate in the synthesis of flavors, fragrances, and pharmaceuticals. It is also employed as a solvent for organic syntheses and can be used in the formulation of insecticides and herbicides.
Safety[edit]
Exposure to 2-pentanol can cause irritation to the eyes, skin, and respiratory system. It is important to handle it with appropriate safety precautions, including the use of personal protective equipment. Ingestion or inhalation of high concentrations can lead to serious health effects, necessitating immediate medical attention.
Environmental Impact[edit]
Like many organic solvents, 2-pentanol has the potential to contribute to air and water pollution if not properly managed. It is biodegradable under aerobic conditions, but its release into the environment should be minimized to prevent potential harm to aquatic life and the broader ecosystem.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
