2-Acetylaminofluorene
2-Acetylaminofluorene[edit]
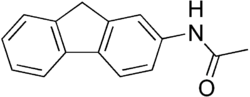
2-Acetylaminofluorene is a synthetic carcinogen that has been widely used in scientific research to study the mechanisms of cancer development. It is an aromatic amine derivative and is known for its ability to induce tumors in various animal models.
Chemical Structure and Properties[edit]
2-Acetylaminofluorene is an aromatic compound with the chemical formula C15H13NO. It consists of a fluorene backbone with an acetylamino group attached to the 2-position. This compound is a pale yellow solid at room temperature and is relatively insoluble in water but soluble in organic solvents.
Mechanism of Action[edit]
The carcinogenicity of 2-Acetylaminofluorene is primarily due to its metabolic activation in the liver. Once ingested, it undergoes enzymatic conversion by cytochrome P450 enzymes to form reactive intermediates. These intermediates can bind to DNA, leading to mutations and the initiation of carcinogenesis.
Use in Research[edit]
2-Acetylaminofluorene has been extensively used in laboratory settings to study the process of chemical-induced carcinogenesis. It serves as a model compound to understand how environmental carcinogens can lead to cancer in humans. Researchers use it to investigate the role of DNA repair mechanisms and the impact of genetic susceptibility on cancer risk.
Health Implications[edit]
Due to its carcinogenic properties, exposure to 2-Acetylaminofluorene is a significant health concern. It is classified as a potential human carcinogen, and safety precautions are necessary when handling this compound in research settings. Understanding its effects has contributed to the development of safety guidelines for handling similar carcinogenic substances.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian