1,3-Butanediol
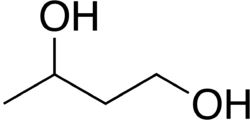
1,3-Butanediol is an organic compound with the chemical formula C₄H₁₀O₂. It is one of the four stable isomers of butanediol. This diol is a colorless, water-soluble liquid with a slightly sweet taste. It is used in various industrial applications, including as a solvent, in the production of plastics, and as a precursor to other chemicals.
Chemical Structure and Properties[edit]
1,3-Butanediol has the structural formula HOCH₂CH₂CH(OH)CH₃. It is a diol, meaning it contains two hydroxyl groups (-OH). The presence of these hydroxyl groups makes it highly soluble in water and gives it hygroscopic properties.
Synthesis[edit]
1,3-Butanediol can be synthesized through several methods, including the hydrogenation of aldol products derived from acetaldehyde and formaldehyde. Another method involves the reduction of 3-hydroxybutanal.
Applications[edit]
Solvent[edit]
1,3-Butanediol is used as a solvent in various industrial processes. Its ability to dissolve both polar and non-polar substances makes it versatile for use in coatings, adhesives, and inks.
Plastic Production[edit]
In the production of polyurethane and other polymers, 1,3-Butanediol serves as a monomer or a chain extender. It contributes to the flexibility and durability of the final product.
Chemical Intermediate[edit]
1,3-Butanediol is a precursor to other chemicals, such as butyrolactone and gamma-hydroxybutyrate (GHB). These compounds have applications ranging from pharmaceuticals to industrial solvents.
Biological Role and Metabolism[edit]
In biological systems, 1,3-Butanediol can be metabolized into beta-hydroxybutyrate, a type of ketone body. This metabolic pathway is of interest in the study of ketogenic diets and metabolic disorders.
Safety and Handling[edit]
1,3-Butanediol is generally considered to be of low toxicity. However, it should be handled with care, as it can cause irritation to the skin and eyes. Proper personal protective equipment (PPE) should be used when handling this chemical.
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
