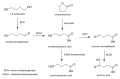
Γ-Butyrolactone
Γ-Butyrolactone (GBL) is a hygroscopic colorless liquid with a weak characteristic odor and profoundly bitter taste. It is a common solvent and reagent in chemistry and is used as an aroma compound, as a stain remover, as a superglue remover, and as a solvent in some wet aluminium electrolyte capacitors.
Chemical Properties[edit]
GBL is a lactone. It is hydrolyzed under basic conditions, for example in a sodium hydroxide solution into sodium gamma-hydroxybutyrate, the sodium salt of gamma-hydroxybutyric acid. Under acidic conditions, it forms an equilibrium mixture of both compounds. These compounds then may go on to form the polymer poly(4-hydroxybutyrate).
Uses[edit]
GBL is not active in its own right; its mechanism of action stems from its identity as a prodrug of gamma-hydroxybutyric acid (GHB). The hypnotic effect of GHB is enhanced by combination with alcohol. A 2003 rat study showed that GBL in combination with ethanol showed a potentiated hypnotic effect, as the sleep-timing measure was longer than both of the individual components combined.
Safety[edit]
Overdose of GBL can cause adverse effects, including sedation, vertigo, nausea, and death. GBL has a distinctive taste and odor, described as being comparable to stale water, synthetic melon aroma, or burnt plastic. This differs significantly from GHB, which is described as having a decidedly salty taste. People who ingest GBL may not be aware they are consuming a drug.
Legal Status[edit]
In many countries, including the United States and Canada, the production, sale, and possession of GBL are illegal since it can be used as a prodrug of GHB. In the United Kingdom, GBL is classified as a Class C drug.
See Also[edit]
Γ-Butyrolactone[edit]
-
Industrial synthesis of gamma-butyrolactone
-
MCPB
-
GHB metabolic pathway
-
GBL jugs
-
Dangerous dietary supplements
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian