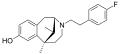
Fluorophen
Fluorophen is a chemical compound that is often used in the field of organic chemistry. It is a derivative of phenol, with a fluorine atom replacing one of the hydrogen atoms in the phenol molecule.
Structure and Properties[edit]
Fluorophen is a monosubstituted phenol, meaning it has one substituent in place of a hydrogen atom on the phenol ring. The substituent in this case is a fluorine atom. The presence of the fluorine atom imparts certain properties to the molecule, such as increased electronegativity and reactivity compared to phenol.
The molecular formula of fluorophen is C6H5FO. It is a colorless liquid at room temperature, with a characteristic aromatic odor. It is soluble in most organic solvents, but only slightly soluble in water.
Synthesis[edit]
Fluorophen can be synthesized by the electrophilic aromatic substitution of phenol with a fluorine source, such as hydrofluoric acid or fluorine gas. The reaction is typically carried out under controlled conditions, as the reaction is highly exothermic and can be dangerous if not properly managed.
Applications[edit]
Fluorophen is primarily used as a building block in organic synthesis. It can be used to prepare a wide variety of fluorinated aromatic compounds, which have applications in fields such as pharmaceuticals, agrochemicals, and materials science.
For example, fluorophen is used in the synthesis of certain fluoroquinolone antibiotics, which are widely used to treat bacterial infections. It is also used in the preparation of fluorinated polymers, which have unique properties such as high thermal stability and resistance to chemical attack.
Safety[edit]
Like many organic compounds, fluorophen should be handled with care. It is a potential skin and eye irritant, and its vapors can be harmful if inhaled. Appropriate safety measures, such as the use of personal protective equipment and adequate ventilation, should be taken when handling this compound.
-
Fluorophen
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian